INCONCRETO NEWS
Digital Disruption in Pharma: Unleashing Innovation and Efficiency
In today’s industrial landscape, digital technology has emerged as a transformative force, revolutionizing traditional processes and operations. From automation to data analytics, digitalization is reshaping how industries operate and innovate, paving the way for enhanced efficiency, productivity, and competitiveness. In April, we delved into the numerous ways digital technology facilitates ongoing industrial transformation, highlighting its symbiotic interplay with industrial processes at every stage of production.
Now, our focus shifts to the Pharmaceutical Industry, exploring the essence of digitization within this sector and its profound implications.
On average, drug companies spend 10 to 15 years developing, validating, and marketing a new product. However, the recent COVID-19 pandemic and successful, lightning-fast mRNA vaccine development shed light on the potential of digital tools to accelerate processes. This major event deepened the pharmaceutical industry’s interest in undergoing digital transformation and implementing cognitive tools into their processes.
Navigating the Digital Frontier: Transformative Applications in Pharma’s Key Domains
François Abiven, CEO of INCONCRETO, outlines five key domains where digital applications are currently making significant strides within the pharmaceutical sector:
- Research and Development (R&D)
Digital technologies are revolutionizing the R&D process in the pharmaceutical industry. Advanced computational methods, artificial intelligence (AI), and machine learning algorithms are being used to analyse vast amounts of data, speeding up the drug discovery process and enabling researchers to identify potential drug candidates more efficiently. Virtual screening, molecular modelling, and predictive analytics are some of the digital tools employed in R&D.
- Clinical Trials
Digital applications are transforming the way clinical trials are conducted. Electronic data capture (EDC) systems, wearable devices, and mobile health (mHealth) technologies are streamlining data collection, enhancing patient monitoring, and enabling remote participation in clinical studies. Digital platforms also facilitate real-time data analysis, improving trial efficiency and reducing costs.
- Industrial Affairs and Manufacturing
Digitalization is optimizing pharmaceutical manufacturing processes and supply chain operations. Internet of Things (IoT) devices, automation, and robotics are being integrated into production facilities to increase efficiency, ensure product quality, and minimize errors. Advanced analytics and predictive maintenance enable proactive equipment monitoring, reducing downtime and enhancing overall productivity.
- Regulatory Affairs
Digital solutions are modernizing regulatory compliance and reporting processes in the pharmaceutical industry. Electronic submission systems, regulatory information management (RIM) platforms, and document management systems facilitate the preparation, submission, and tracking of regulatory documents and filings. Digital platforms also support compliance with evolving regulatory requirements, ensuring adherence to quality standards and guidelines.
- Sales
Digital technologies are reshaping pharmaceutical sales and marketing strategies. Customer relationship management (CRM) systems, digital detailing platforms, and data analytics tools are enabling pharmaceutical companies to personalize their interactions with healthcare professionals (HCPs) and target audiences. Digital marketing campaigns, social media engagement, and online educational resources are also being utilized to enhance brand awareness and product promotion.
Overall, digital applications across these five domains are driving innovation, improving operational efficiency, and accelerating the pace of development and commercialization in the pharmaceutical sector. Notably, the pharmaceutical industry has witnessed a surge in digital investments, particularly accelerated by the Covid-19 pandemic. Among these domains, R&D and clinical trials stand out for their advanced deployment of digital applications.
Unlocking Biopharma’s Digital Potential: From Clinical Trials to Manufacturing Efficiency
L.E.K. Consulting’s recent work with pharmaceutical clients globally shows there is significant potential to be found by looking beyond multichannel offers. Return on investment in excess of a 20% EBITDA uplift can be achieved through digital transformation in other parts of the value chain, including production planning, inventory management, operational efficiency, and customer management and engagement.
L.E.K surveyed 65 biopharmaceutical companies around the globe as part of their recent digital excellence benchmarking survey, September 2023, approximately half the respondents admitted to having no real digital strategy (see Figure 1). Encouragingly, 60%-75% of companies were confident that in the midterm they would have advanced digital capabilities in place across multiple areas.
A glance at the global pharmaceutical industry digitalisation picture shows Europe at a level comparable to that of the US and Latin America. However, ambitions for the future are similar across geographies. Where differences can be seen is in identifying what has been prioritised over the past three to five years. European and US biopharma companies have placed a huge emphasis on drug discovery and clinical development, with distribution and portfolio/corporate strategy being the lowest investment priorities. Looking to the future, European companies also indicated a likely increase in investment in downstream processes (see Figure 2).
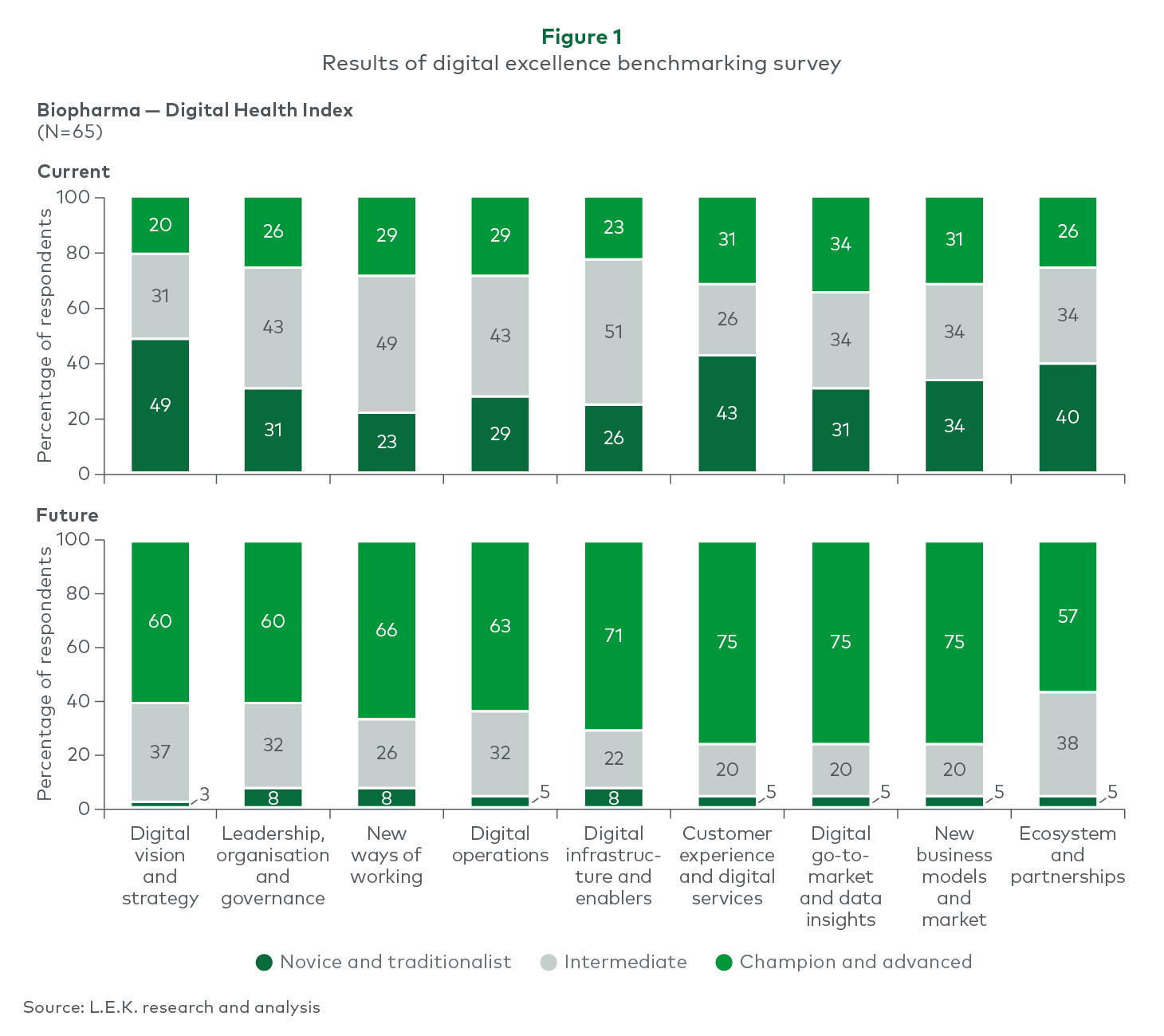
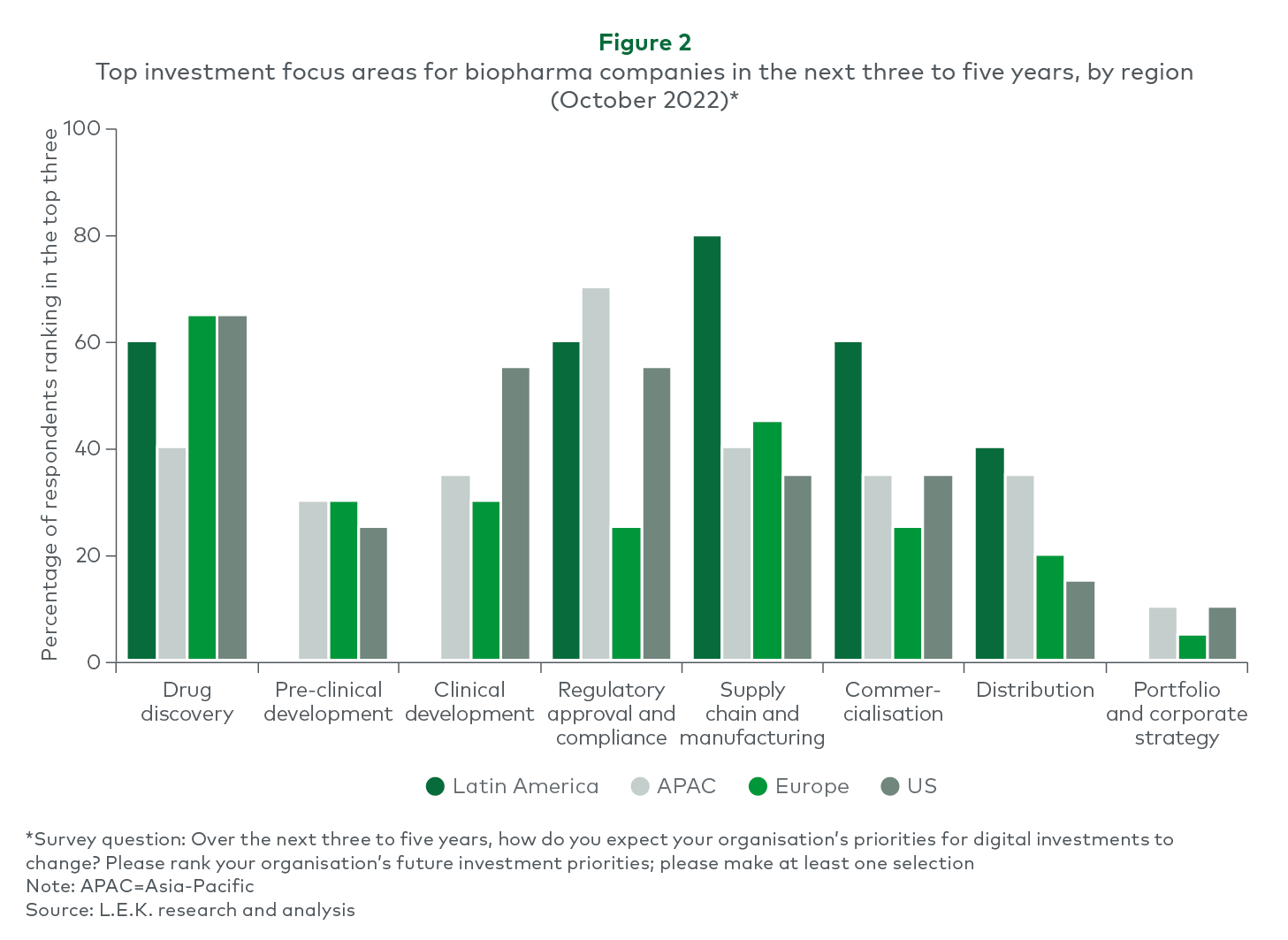
Interestingly, the National Center for Biotechnology Information reveals that various digital health technologies have experienced evaluation in clinical development programs, including external, wearable, implantable, and ingestible devices and sensors, as well as digital mobile health applications accessible via personal electronic devices like smartphones, tablets, and computers. Some of these technologies have been integrated into ongoing neurology and respiratory clinical development programs.
The primary advantage of digital health technologies lies in their capacity to continuously collect objective or biological data outside of office visits, enhancing the efficacy and safety information available compared to traditional episodic, time point-based data capture.
Digital and analytics offer the promise of revolutionizing biopharma manufacturing, leading to significant improvements in quality, speed, agility, and resilience. Already, leading players have leveraged these tools to achieve remarkable performance gains, including increased plant capacity, reduced lead times, and lowered conversion costs. However, realizing the full potential of digital transformation requires a bold reimagination of manufacturing operations.
McKinsey’s article “Reimagining the future of biopharma manufacturing”, published in October 2022, explain us that successful digital transformation in biopharma typically follows one of three paths: implementing use cases to address specific pain points, developing lighthouse sites for holistic transformation, or undertaking network-wide transformation across all operations.
To embark on this digital journey, biopharma companies must set bold ambitions aligned with their strategic goals. This involves defining the desired impact of digital transformation, determining the scale of implementation, establishing leadership and governance structures, investing in technology and data infrastructure, and fostering a culture of digital innovation.
Innovative Digital Strategies Transforming Pharmaceutical Manufacturing
Large pharmaceutical groups like Sanofi and GSK are leading the charge, implementing innovative digital solutions to enhance efficiency, quality, and agility in their industrial operations.
Sanofi’s Smart Facilities:
Sanofi, a global pharmaceutical giant, has embarked on a journey to develop and launch smart facilities that harness the power of digitalization. In partnership with industry leader Dassault Systèmes, Sanofi has been pioneering the concept of smart manufacturing in France and Singapore. These state-of-the-art facilities integrate advanced digital technologies, such as artificial intelligence, Internet of Things (IoT), and data analytics, to optimize production processes and drive operational excellence.
Through the implementation of digital twins – virtual replicas of physical manufacturing environments – Sanofi is able to simulate and analyse various scenarios in real-time, enabling proactive decision-making and predictive maintenance. This predictive capability not only minimizes downtime but also ensures optimal resource utilization and product quality.
GSK’s Digital Manufacturing Initiatives:
Meanwhile, GSK, another leading player in the pharmaceutical industry, has been making significant strides in digital manufacturing. With a focus on harnessing digital technologies to streamline production processes and enhance product quality, GSK has implemented a range of innovative initiatives across its manufacturing network.
One notable advancement is GSK’s adoption of advanced analytics and machine learning algorithms to optimize manufacturing operations. This data-driven approach not only enhances operational efficiency but also ensures product consistency and compliance with regulatory requirements.
Furthermore, GSK is leveraging digital solutions to enable remote monitoring and management of manufacturing facilities, providing real-time visibility into production activities and performance metrics. This remote monitoring capability enhances agility and responsiveness, allowing GSK to quickly address any issues or deviations that may arise during production.
Navigating the Path to Digital Transformation in Pharma
According to CAS, a Division of the American Chemical Society, about 55% of pharmaceutical firms report using digital technologies to some degree. However, a lack of expertise in knowledge management and experience with digital tools often transforms this smart initiative into a debatable investment. With roughly 70% of digitalization programs failing, pharma companies need to reevaluate where to invest their digitalization dollars and optimize their deployment strategies to unlock competitive advantages and generate life-changing pharmaceuticals.
With a deep understanding of robust knowledge management, cognitive tools, and how they intertwine, pharma companies can revolutionize their processes at all levels and breed better global healthcare.
As pharma’s digital transformation continues to evolve, digital technologies and cognitive tools are finding their way into all aspects of the industry, allowing for faster drug development and expanded treatment options for a growing number of conditions. Digital transformation strives to bring innovative healthcare solutions in a sustainable, responsible, and accessible way.
Discover the expertise of INCONCRETO and its Partners in digital transformation projects, tailored specifically for the healthcare and pharmaceutical industry.
Connect with our team to leverage our technical proficiency and extensive experience in large-scale program execution. With a focus on improving predictability and maximizing profitability, we specialize in guiding Capex and Opex project investments to achieve optimal outcomes in pharmaceutical ventures.
For further readings, you may consult these sources
https://exa4mind.eu/application-cases
https://www.lek.com/insights/hea/eu/ar/digital-transformation-pharmaceutical-industry
https://www.cas.org/resources/cas-insights/digital-rd/digital-transformation-pharma
https://www.ia-grp.com/views/digital-clinical-trials-imaging/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9341314/
https://www.fiercepharma.com/manufacturing/gsk-opens-doors-110m-biologics-smart-factory-uk
https://www.multivu.com/players/English/8627651-sanofi-new-digital-manufacturing-facility
Newsletter
© INCONCRETO. All rights reserved. Powered by AYM













































